Crystal Structures Reveal Hydrogen-Bonding Patterns in Concentrated Aluminate Solutions
The size of the alkali metal influences the crystallization pathway
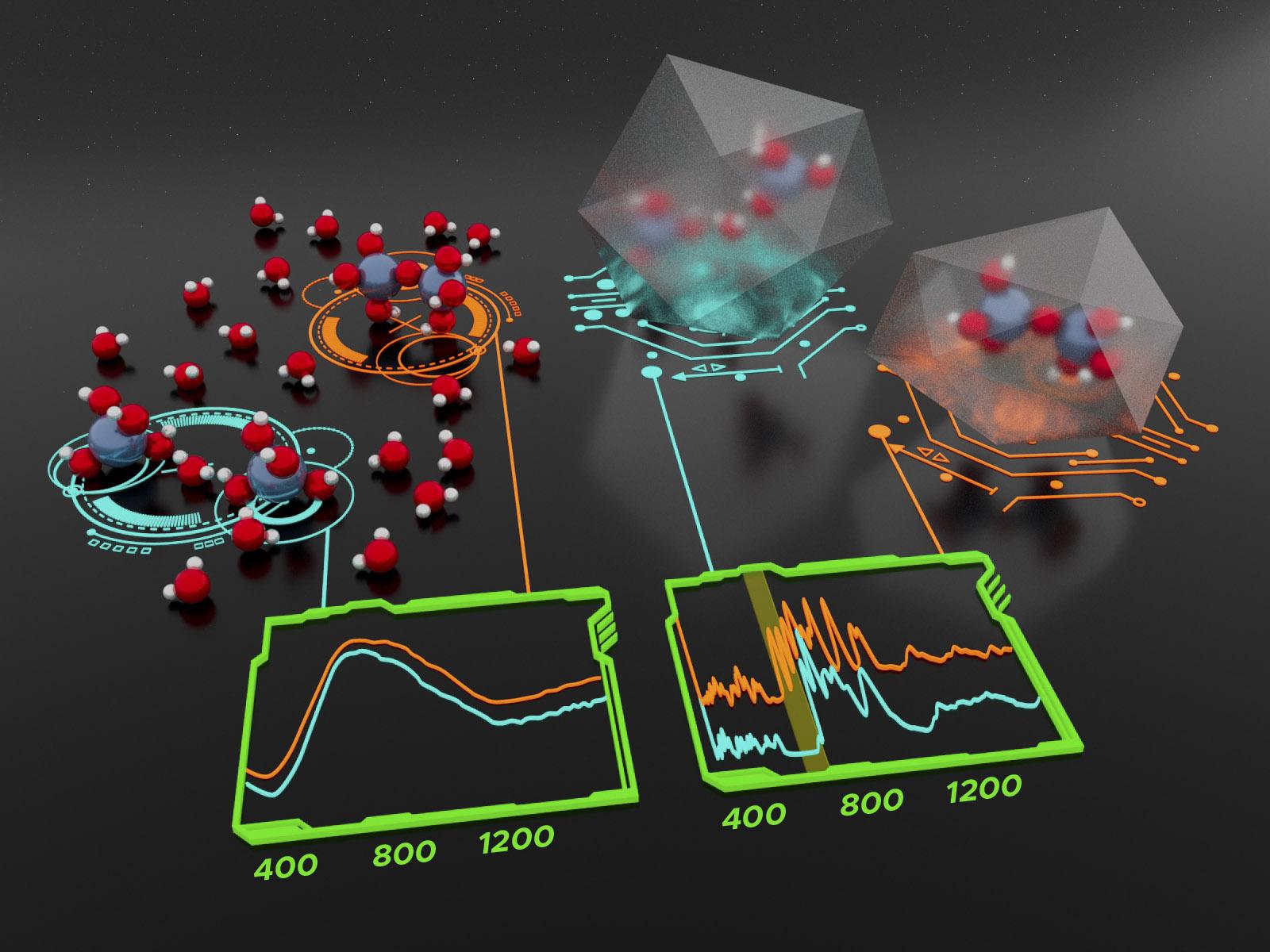
Competition between hydrogen bonding involving water and aluminate groups and steric effects is reflected in the cation size dependence of the crystal structure of alkali aluminate salts. This difference is manifested in signatures in the inelastic neutron scattering spectrum that shed light on the bonding behavior of aluminate ions in solution.
(Illustration by Cortland Johnson | Pacific Northwest National Laboratory)
The Science
In concentrated alkaline solutions, aluminate ions can form either monomeric, Al(OH)4-, or dimeric, Al2O(OH)62-, species. Researchers at the Interfacial Dynamics in Radioactive Environments and Materials (IDREAM) Energy Frontier Research Center (EFRC) utilized inelastic neutron scattering (INS), Raman spectroscopy, and ab initio calculations based on density functional theory (DFT) to understand this speciation. They synthesized crystalline aluminate salts with different alkali metals—either potassium (K), rubidium (Rb), or cesium (Cs)—and a sodium (Na) aluminate solution and compared their spectra. They found that the size of the alkali metal controls the formation of hydrogen bonds and influences the crystal structure of the salts. The synthesized salts, which had well-defined crystal structures, allowed the relationship between cation size and hydrogen bonding to be interrogated without the confounding effects of random solution motion, shedding light on the dynamics in more complicated solutions.
The Impact
Understanding aluminum speciation has important applications in the retrieval, processing, and vitrification of caustic nuclear waste, like that stored at the Hanford Site. Uncertainties in aluminum solubility make it difficult to predict how aluminum minerals in nuclear waste will change following dilutions or additions during waste retrieval and processing. This study provides insight into the speciation and crystallization of aluminum-based salts, especially the energetic contribution of hydrogen bond networks around solvated metal ions. Additionally, the knowledge from this study can be applied to other concentrated electrolyte systems in energy related applications, such as catalysis and energy storage.
Summary
Researchers collected INS and Raman spectra of K, Rb, and Cs aluminate salts and a frozen Na-aluminate solution. They also simulated these spectra through DFT calculations. Differences in the spectra can be attributed to the hydrogen-bonding network of the salts. The results indicate that the larger radius of the Cs cation may hold the aluminate crystal framework open to allow water to enter the structure and complete the hydrogen bond network. Thus, the difference in crystallization patterns can be attributed to the size of the cation. These results also indicate that INS can be used as a sensitive probe for the strength and completeness of hydrogen bonds in different materials.
INS was performed at Oak Ridge National Laboratory’s Spallation Neutron Source, supported by the Department of Energy (DOE), Office of Basic Energy Sciences, Scientific User Facilities Division. Raman spectroscopy was performed using facilities at the Environmental Molecular Science Laboratory, a DOE Office of Science user facility.
Contact
Micah Prange
Computational Scientist, Physical and Computational Sciences Directorate
micah.prange@pnnl.gov
Carolyn Pearce
Director, IDREAM
carolyn.pearce@pnnl.gov
Funding
This research was supported by the IDREAM EFRC.
Published: June 6, 2022
Prange, M.P., T.R. Graham, R. Gorniak, M. Pouvreau, M. Dembowski, H.-W. Wang, L.L. Daemen, G.K. Schenter, M.E. Bowden, E.T. Nienhuis, K.M. Rosso, A.E. Clark, and C.I. Pearce. 2021. “Theory-Guided Inelastic Neutron Scattering of Crystalline Alkaline Aluminate Salts Bearing Principal Motifs of Solution-State Species.” Inorganic Chemistry 60 (21): 16223–16232. American Chemical Society (ACS). https://doi.org/10.1021/acs.inorgchem.1c02006.
Dembowski, M., M.P. Prange, M. Pouvreau, T.R. Graham, M.E. Bowden, A. N’Diaye, G.K. Schenter, S.B. Clark, A.E. Clark, K.M. Rosso, and C.I. Pearce. 2020. “Inference of principal species in caustic aluminate solutions through solid-state spectroscopic characterization.” Dalton Transactions 49 (18): 5869–5880. https://doi.org/10.1039/d0dt00229a.