Discovering New Kinds of Crystal Defects
IDREAM researchers unearth new defects during gibbsite crystallization
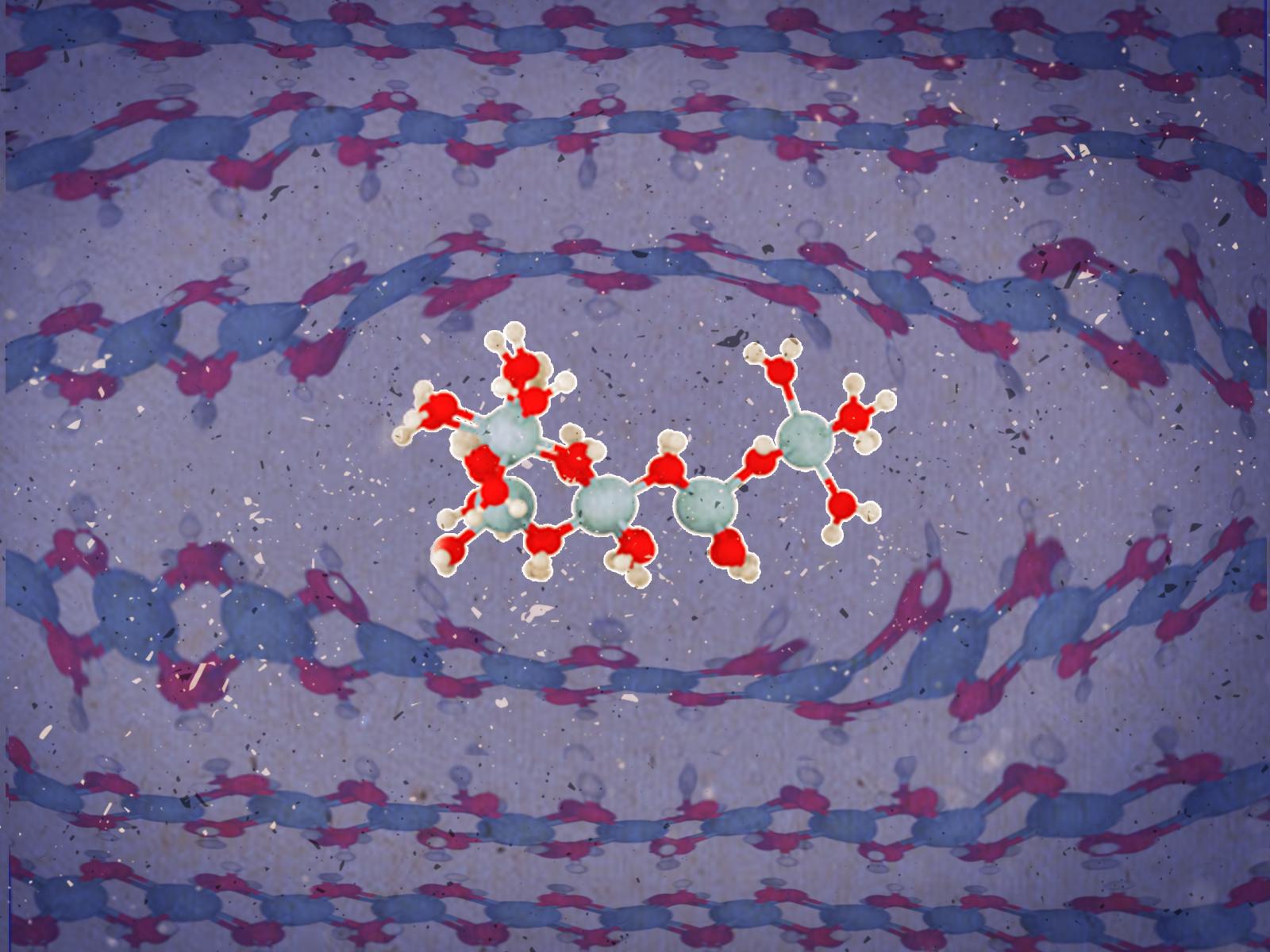
Rapid crystallization of amorphous aluminum hydroxide gel traps smaller clusters between layers, resulting in defects in the crystal. Under alkaline conditions, gibbsite grows by more ordered layers.
(Illustration by Stephanie King | Pacific Northwest National Laboratory)
The Science
Gibbsite is a common aluminum-bearing mineral in the environment and in industrial applications. However, the growth mechanism of its layered structure is poorly understood. Researchers from the Interfacial Dynamics in Radioactive Environments and Materials (IDREAM) Energy Frontier Research Center (EFRC) synthesized gibbsite nanoplates under acidic to neutral conditions and analyzed their structure. They then followed the evolution of the structure as it grew under alkaline conditions. The researchers determined that when gibbsite is synthesized at acidic to near-neutral conditions, the material contains misaligned and truncated sheets. This conclusion is consistent with the observation that rapid crystallization of the amorphous aluminum hydroxide gel traps smaller clusters between layers. The researchers also showed that gibbsite grows by more ordered layers under alkaline conditions, preserving the truncated sheets from more acidic conditions.
The Impact
Understanding how gibbsite forms and reacts with its surroundings is important for many environmental and industrial applications, including the treatment of millions of gallons of legacy high-level radioactive waste stored in underground tanks at Department of Energy sites. This research provides insight into the differences in gibbsite reactivity under different growth conditions. The researchers propose that increased reactivity is likely related to the large-scale structural defects caused by the incorporated solution clusters and mis-stacked gibbsite layers. This information can be used to generate well-characterized gibbsite nanoplates at a large scale for cross-EFRC distribution and additional research to unravel the influence of gibbsite structure on its chemical reactivity.
Summary
Using a multi-instrument approach, including scanning electron microscopy, magic-angle spinning nuclear magnetic resonance, Raman and infrared spectroscopies, X-ray diffraction, and X-ray total scattering pair distribution function analysis, IDREAM researchers characterized gibbsite nanoplates synthesized at low pH. They then followed the evolution of the structure during its reaction under highly alkaline conditions at 80 °C. Characterization revealed that the best structural fit for the initial nanoplates required the inclusion of encapsulated clusters. These defects are preserved within the particles during aging and growth at high temperature.
Results from this study suggest complex aluminum speciation in solution at low pH, even in the presence of the more stable gibbsite phase. Further, these flexible defect structures may sorb and subsequently entrain other elements in the growing gibbsite particles, affecting their overall reactivity. This research was conducted at Pacific Northwest National Laboratory and the Advanced Photon Source, operated by Argonne National Laboratory, where data acquisition was performed at beamline 11-ID-B.
Contact
Sebastian Mergelsberg
Chemist, Physical and Computational Sciences Directorate
sebastian.mergelsberg@pnnl.gov
Carolyn Pearce
Director, IDREAM
carolyn.pearce@pnnl.gov
Funding
This research was supported by the IDREAM EFRC.
Published: April 8, 2022
S.T. Mergelsberg, M. Dembowski, M.E. Bowden, T.R. Graham, M.P. Prange, H.W. Wang, X. Zhang, O. Qafoku, K.M. Rosso, C.I. Pearce. Cluster defects in gibbsite nanoplates grown at acidic to neutral pH, Nanoscale 2021. [DOI: 10.1039/D1NR01615F]