Technology Overview
Solution for Longer Seasonal Energy Storage
Wind and solar power generation are on the rise as an abundant source of energy, but it is intermittent by nature and cannot be accessed on demand. Mechanisms are needed to store this energy long-term so that it can be made available whenever needed to bolster the power grid.
Pumped hydro storage is the most used long-term energy storage mechanism across the globe, but power distribution is limited due to regulatory concerns and geological constraints. Conversely, green hydrogen generated by excess electricity from renewable energy is a possible solution for seasonal storage, but it is hampered by hydrogen storage challenges and low roundtrip efficiency—the amount of energy put into storage that can later be retrieved. Finally, conventional batteries are not a recommended solution because of self-discharge, limiting storage to only days or weeks.
Researchers at the Pacific Northwest National Laboratory (PNNL) have developed a solution for long-term, seasonal energy storage. The innovative Temperature-based Hibernating Battery effectively cuts off the self-charge function during the battery’s idling period, making it ideal for long-term electricity storage without loss of electricity for several months.
Temperature Rising
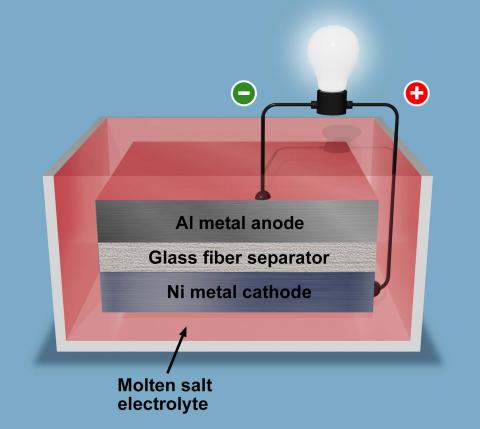
The Temperature-based Hibernating Battery consists of a metal anode, a metal cathode, a molten salt electrolyte, and a porous separator.
The battery operates only at an elevated temperature, at which the molten salt electrolyte is in a liquid state. When the battery is idle, it is kept at ambient temperature to avoid self-discharge—effectively “freezing” the electrolytes.
During the charging process, the temperature of the battery is raised by an external heat source, such as a heating cartridge installed on an industrial-scale system, to bring the electrolytes to a liquid state to initiate ion mobility in the battery.
Once the battery is fully charged, the battery is then cooled to the ambient temperature, allowing the electrolyte to solidify—keeping the battery at an all-solid state. Because the solidified electrolyte does not have ion transport capability, self-discharge of the battery can be cut off while at ambient temperature.
When the battery reaches the optimum operating temperature, the battery discharges, providing energy to the grid.
The battery is configured with the metal anode and cathode sandwiched between a glass fiber separator. All are immersed in a molten electrolyte. The electrolyte is heated up into liquid, with a conventional glass fiber separator mitigating thermal expansion of the solidification and insulating the cathode from the anode. Once the battery is not being used or charged, the molten electrolyte cools at ambient temperature into solid salts.
Candidate metals for the anode include aluminum, magnesium, titanium, and lead, all with lower electrochemical redox potential. For the cathode, nickel, zinc, iron, and copper have higher electrochemical potential.
Industry Applications
Grid operators and utilities who deploy batteries for long-duration energy storage purposes would benefit from this self-charging, heat-activated battery technology.
Available for Licensing
This technology is available for licensing. You can view all of PNNL’s market-changing intellectual property at pnnl.gov/available-technologies.
Advantages
- Low-cost, long-term storage of renewable energy for months or years.
- No geological constraints or regulatory concerns that typically hamper pumped storage hydro.
- Low self-discharge mechanism compared to conventional batteries.
- Higher roundtrip efficiency than green hydrogen.
- Cycling stability supports seasonal storage.
- Simple cell architecture.
- High safety, with low fire hazard and easy transportation.