Directly Measuring X-Ray Enhanced Dissolution of Gibbsite
Irradiation in a sodium hydroxide solution increases the dissolution rate and particle roughness of gibbsite compared to dry irradiation
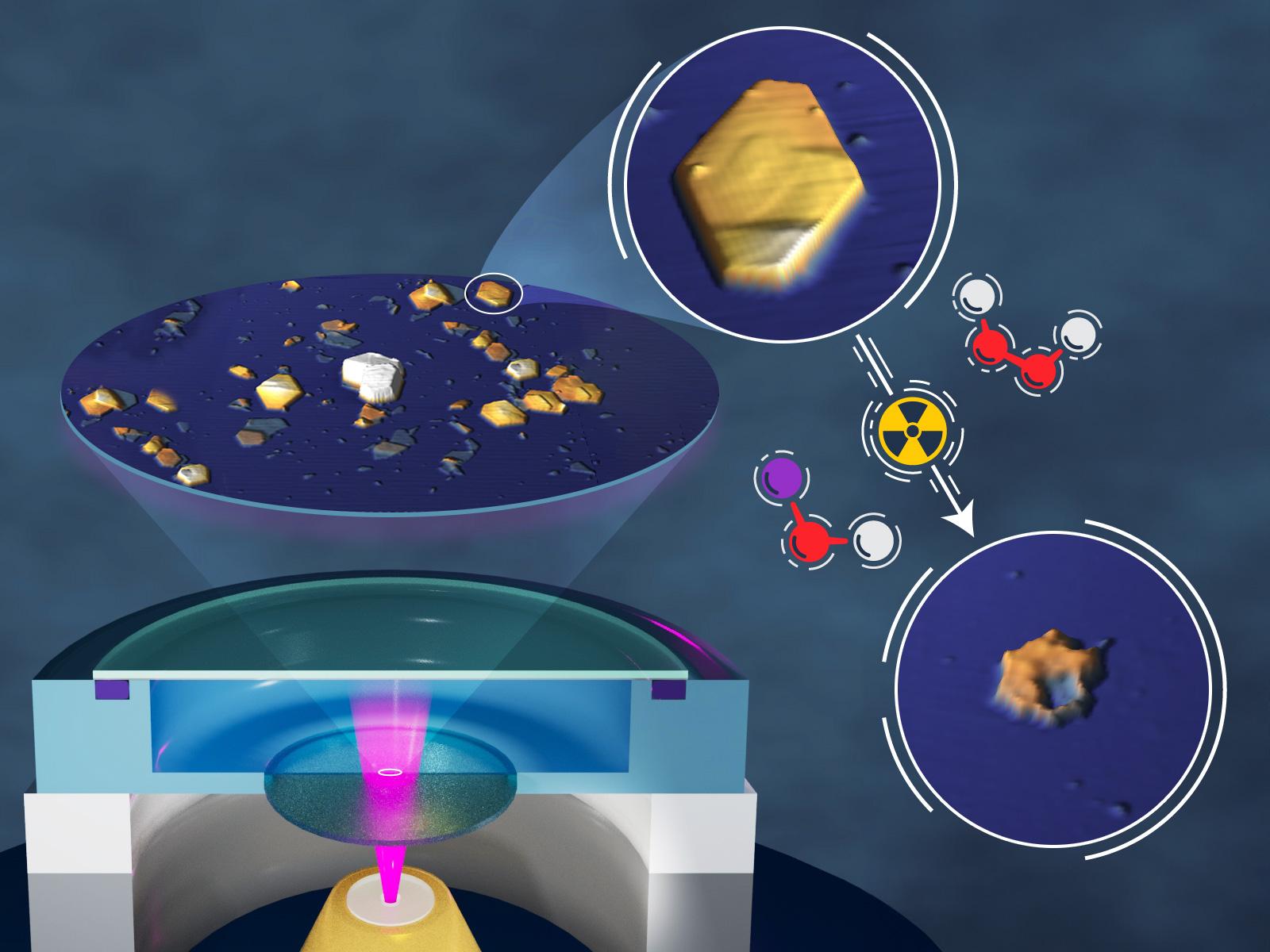
Whether gibbsite is dry or wet, irradiation affects how it dissolves.
(Image by Nathan Johnson | Pacific Northwest National Laboratory)
The Science
Radiation has a myriad of effects on materials and solutions, which can complicate chemical behavior. This makes developing an understanding of processes like precipitation, aggregation, and dissolution challenging. Researchers studying the dissolution of gibbsite particles in sodium hydroxide (NaOH) explored the difference in the dissolution rate and particle roughness when gibbsite was irradiated dry or in solution. They found a significant enhancement in the dissolution rate and roughness when the particles were irradiated during dissolution in NaOH compared to particles irradiated prior to dissolution. This can likely be attributed to the combined effects of irradiation on both the particles themselves and the solution, which produces reactive species in the solution.
The Impact
Predicting and manipulating the aggregation and precipitation of minerals in the presence of radiation is extremely challenging given the complexities of the multiple, intersecting processes. However, it is critical to store, extract, and remediate nuclear wastes, including legacy tank waste at the Hanford site. Aggregates can change the behavior of waste solutions, potentially causing problems during processing. This work points to differences in how radiation can affect gibbsite dissolution based on when irradiation occurs and the conditions during irradiation. Identifying the combined effects of radiation on the gibbsite and surrounding solution is important to developing an accurate understanding of mineral dissolution under extreme conditions.
Summary
The stability of minerals in extreme and changeable environments, such as highly concentrated solutions and under irradiation, is challenging to predict. It is particularly important for understanding the behavior of legacy radioactive tank waste that needs to be processed. A team of researchers studied the dissolution behavior of irradiated gibbsite, an aluminum-based mineral, under different conditions. They compared the dissolution rate and surface roughness of gibbsite irradiated dry or while in a solution of NaOH, as well as the relevant control samples. The team found that both the dissolution rate and particle roughness were enhanced when the samples were irradiated. This was particularly notable for the particles irradiated in the NaOH solution, which showed significant increases in dissolution rate and roughness compared to all other samples. The enhancement likely comes from the combined effects of radiation on both the solution and the gibbsite. The hydroxide in solution can convert to highly reactive peroxides that cause further reactions on the gibbsite surface. This work provides a fundamental understanding of the how radiation can affect critical processes in materials.
PNNL Contact
Carolyn Pearce, Pacific Northwest National Laboratory, carolyn.pearce@pnnl.gov
Shawn Riechers, Pacific Northwest National Laboratory, shawn.riechers@pnnl.gov
Funding
This research was supported by Interfacial Dynamics in Radioactive Environments and Materials (IDREAM), an Energy Frontier Research Center funded by the Department of Energy, Office of Science, Basic Energy Sciences (FWP 68932). A portion of the research was performed in EMSL, the Environmental Molecular Sciences Laboratory, a national scientific user facility at PNNL which is sponsored by the DOE Biological and Environmental Research program. PNNL is a multiprogram national laboratory operated for DOE by Battelle Memorial Institute under contract no. DE-AC05-76RL0-1830.
Published: January 2, 2024
Riechers, S. L., X. Zhang, J. A. LaVerne, K. M. Rosso. 2023. “Direct Measurement of Mineral Dissolution Enhanced by X-ray Irradiation.” J Phys. Chem. C., 127 (36), 17694-17699. [DOI: 10.1021acs.jpcc.3c02817]