When Less Is More: Fewer Proton Relays Improve Catalytic Rates
First direct comparison of three nickel-based complexes shows complexes with 2 proton relays outperform those with 4
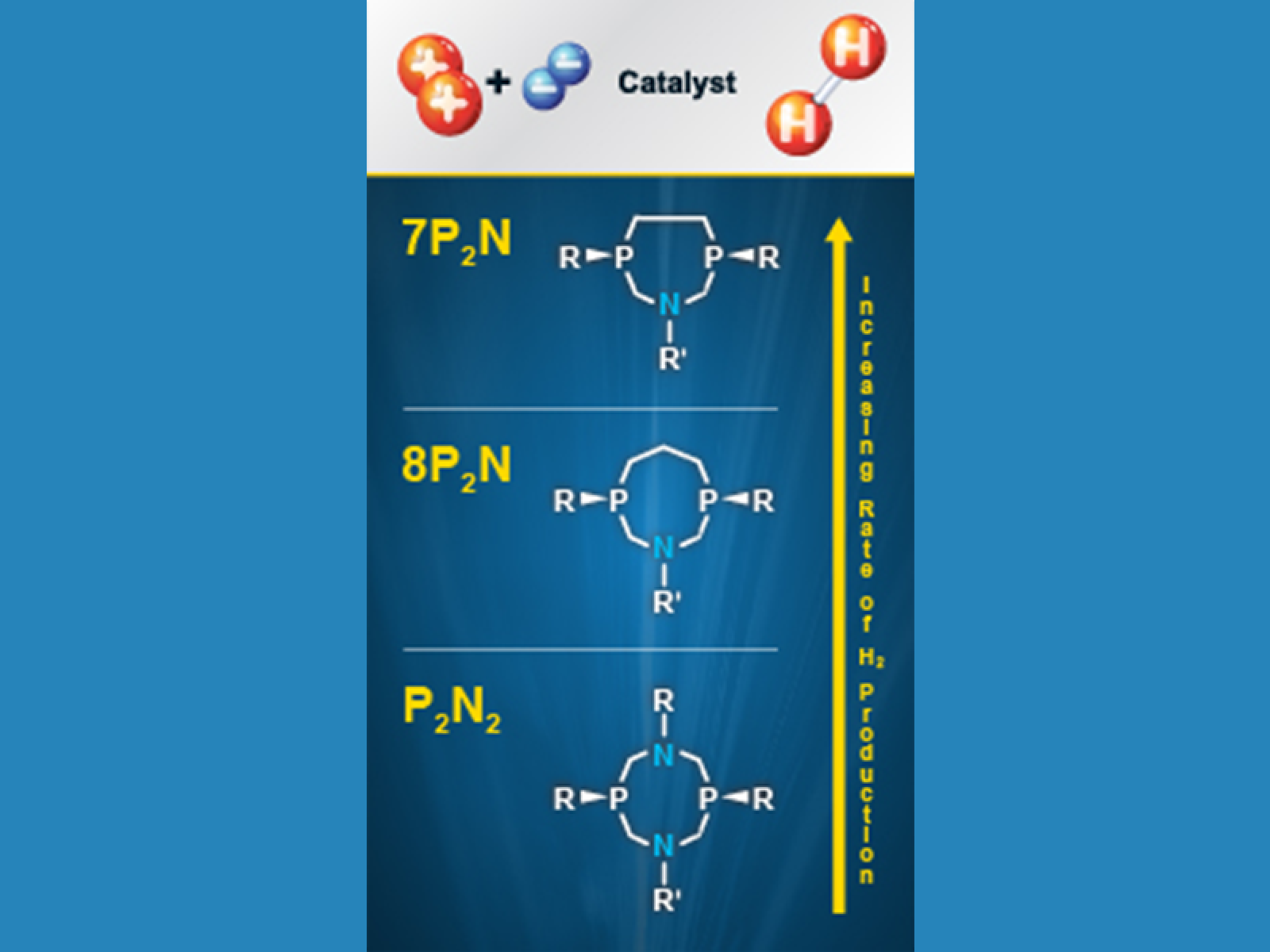
With just two proton relays or pendant amines and a smaller ring structure, nickel-based catalyst with a 7P2N ligand is faster than two similar catalysts, according to a detailed comparison performed by scientists at the Center for Molecular Electrocatalysis.
Results
By directly comparing three closely related catalysts, scientists at the Center for Molecular Electrocatalysis established that hydrogen production speed and efficiency are influenced by the molecules' structure and proton relay arrangement, not the total number relays. They found that a catalyst built with the ligand called 8P2N, with at total of two proton relays in its nickel complex, is faster than a catalyst with double the number. Further, catalysts built with the related 7P2N, with its smaller heptagonal ring of atoms, is faster than its 8P2N counterpart, with a larger octagonal ring.
"We don't necessarily need more proton relays to improve catalytic performance," said Dr. Monte Helm, Deputy Director, Center for Molecular Electrocatalysis. "This work demonstrates that delivering protons to the right spots for the catalyst to use productively is the most important factor in determining catalytic performance."
The Center is an Energy Frontier Research Center, funded by the U.S. Department of Energy's Office of Basic Energy Sciences and led by Pacific Northwest National Laboratory.
Why it matters
Storing energy from wind farms and other renewable sources in chemical bonds relies on platinum-based catalysts, which excel at turnover frequency and overpotential. Turnover frequency is the rate at which the desired molecules are created. Overpotential, the difference between the theoretical energy and the actual energy used, measures the reaction's efficiency. Because platinum is scarce, a catalyst based on earth-abundant metals is needed to store large volumes of renewable energy and release it when needed. This study sheds light on protons' behavior in such catalysts, key to synthesizing catalysts that can match or beat platinum's high turnover frequency and low overpotential.
"Right now, the catalyst used to store energy in chemical bonds with fuel cells is platinum, which we can't implement on a large scale because of cost and availability," said Helm. "To increase the use of renewable sources of energy, we need a way to store the energy during times of high production to balance those times of low production."
Methods
The team began with three nickel-based electrocatalysts, which are working models of hydrogenase enzymes that split and form hydrogen for water-dwelling microbes. The models allowed for two vital comparisons:
- Comparing proton relays. The team compared catalysts with P2N2 and with 8P2N ligands; the catalysts are identical except for the number of proton relays. Nickel complexes with P2N2 have four relays, and those with 8P2N have two.
- Comparing structure. The team compared catalysts with 8P2N and 7P2N ligands; these catalysts differ only in the size of the rings that contain the proton relays. 8P2N has an eight-membered cyclic ligand, while 7P2N has a smaller seven-membered cyclic ring.
In doing the two comparisons, the team found that the nickel catalyst with P2N2 was the slowest, generating 1,040 molecules of hydrogen per second with an overpotential of 570 mV. The nickel catalyst with 8P2N achieved a maximum turnover frequency for H2 production of 3,300 molecules/second with an overpotential of 760 mV. The nickel catalyst with 7P2N achieved a maximum turnover frequency for hydrogen production of 17,000 molecules/second with an overpotential of 860 mV.
"The turnover frequency of the nickel complex with 8P2N is limited by the formation of catalytically less-productive protonated forms of the complex," said Dr. Simone Raugei, who performed the computational studies for the team. "These forms divert the catalyst away from its work, slowing it down."
With a smaller ring that allowed it to better position the protons for the reaction, 7P2N clocked in at 17,000 molecules/second with an overpotential of 860 mV. "This is good news, but we want to know more," said Helm. "Our focus now is to understanding how to make catalyst modifications that will lower the overpotentials while maintaining the fast rates."
What's next?
The team at the Center for Molecular Electrocatalysis is continuing to answer fundamental questions about the behavior of protons in reactions important to energy storage and production. Look for follow-on work on the electronic nature in the catalytic complexes that control the energy of reaction intermediates as the team works to uncover more ways to ratchet down the overpotential.
Sponsor
Center for Molecular Electrocatalysis, an Energy Frontier Research Center funded by the U.S. Department of Energy, Office of Science, Basic Energy Sciences
Research team
Stefan Wiese, Uriah J. Kilgore, Ming-Hsun Ho, Simone Raugei, Daniel L. DuBois, R. Morris Bullock, and Monte L. Helm, PNNL
Published: January 1, 2014
Wiese S, UJ Kilgore, MH Ho, S Raugei, DL DuBois, RM Bullock, and ML Helm. 2013. "Hydrogen Production Using Nickel Electrocatalysts with Pendant Amines: Ligand Effects on Rates and Overpotentials." ACS Catalysis 3(11):2527-2535. [DOI: 10.1021/cs400638f]