Simulating the Global Distribution and Climate Impacts of Nitrate Aerosol
Advanced atmospheric aerosol treatment in a climate model enables assessments of the radiative effects of nitrate aerosols
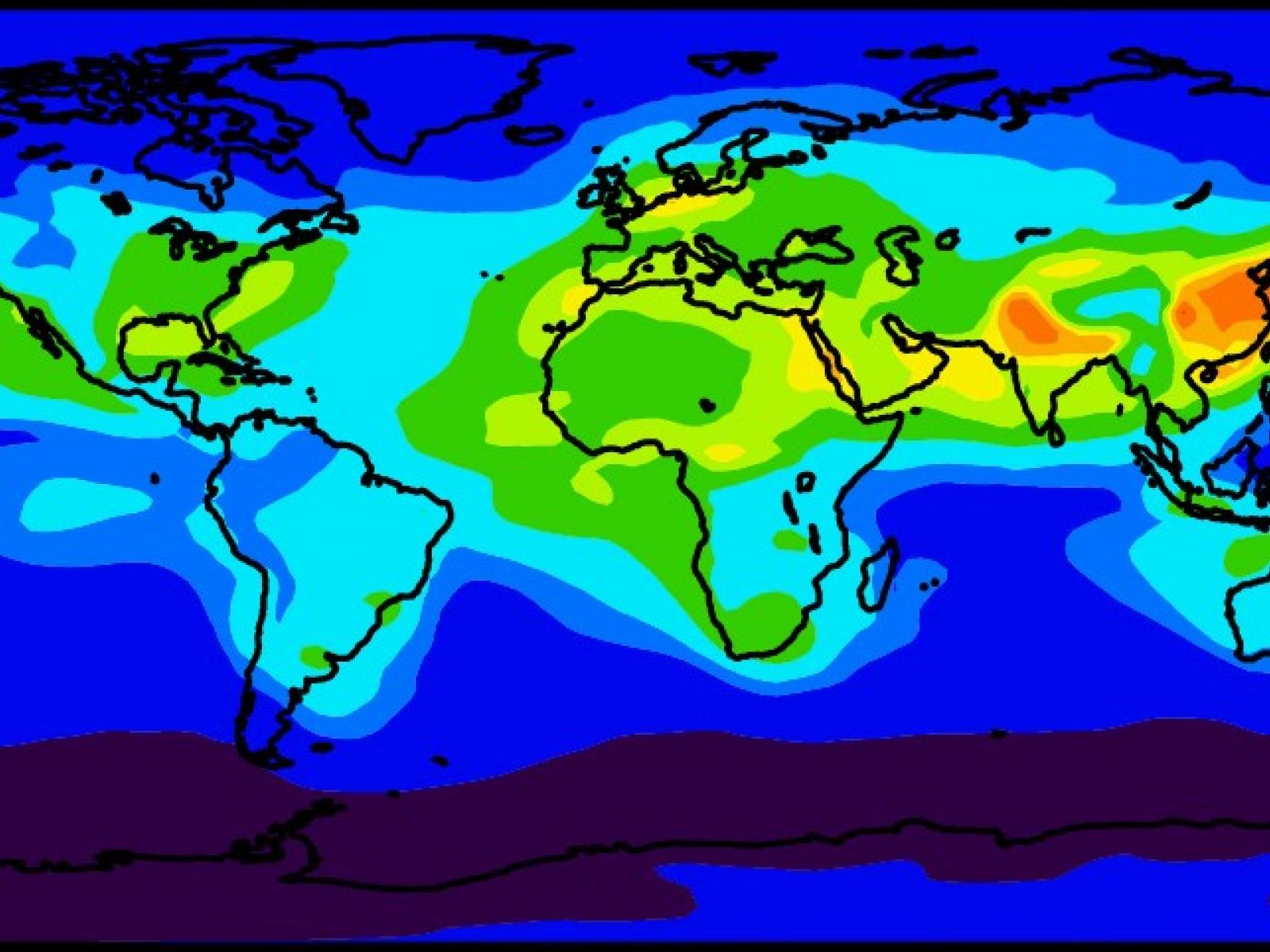
Nitrate aerosols form globally via the atmospheric oxidation of nitrogen oxides emitted in large quantities from natural sources and current human activities.
(Image by Rahul Zaveri | Pacific Northwest National Laboratory)
The Science
Atmospheric aerosols modulate Earth’s energy balance both directly by scattering and absorbing incoming solar radiation and indirectly by forming clouds that reflect radiation back to space. Ammonium nitrate, which forms when ammonia combines with the nitric acid vapors produced from atmospheric oxidation of nitrogen oxides, composes a notable fraction of aerosols. Researchers introduced an advanced aerosol chemistry module to a climate model, enabling it to simulate the formation and radiative impacts of nitrate aerosol. They found that incorporating nitrate formation increases global average aerosol number concentrations in the size range that scatters solar radiation and seeds clouds by about 10%, leading to an appreciable cooling effect that partially offsets greenhouse gas warming.
The Impact
Aerosols and aerosol–cloud interactions remain the largest uncertainty in assessing climate change. While there are natural sources of the base materials of nitrate aerosols, human activity during the industrial era has significantly increased their emissions. This makes understanding how human-caused emissions change Earth’s natural radiative balance crucial. The advanced aerosol-climate model developed in this study reliably captures the complex dynamics of nitrate aerosol formation. This new model will enable more accurate predictions of nitrate aerosol formation from various pollution sources across the world and their influence on climate change.
Summary
Submicron-sized atmospheric aerosols, typically composed of sulfate and nitrate salts, are particularly crucial in modulating the Earth’s radiative balance. Sulfate and nitrate aerosols are very efficient at scattering solar radiation as well as facilitating cloud formation. Both aerosols largely form in the atmosphere via gas-to-particle conversion of the sulfur dioxide, nitrogen oxides, and ammonia released in large quantities from a variety of natural and human sources. Substantial work has focused on sulfate, but developing a faithful numerical representation of nitrate aerosol in climate models has been difficult because of the semivolatile nature of ammonium nitrate.
In this study, researchers introduced the advanced aerosol chemistry module—Model for Simulating Aerosol Interactions and Chemistry—into a state-of-the-science global climate model—Community Atmosphere Model, version 5—to properly simulate the complex dynamics of submicron ammonium nitrate formation and its chemical interactions with naturally occurring sea salt and dust aerosols. Nitrate inclusion resulted in an approximately 10% increase in the global average number concentrations of aerosols in the key size range for efficient interactions with solar radiation and seeding cloud droplets. Consequently, nitrate aerosols accounted for about −0.7 W m-2 of radiative cooling. The numerical simulations were performed using the computational resources at the Environmental Molecular Sciences Laboratory, a U.S. Department of Energy Office of Science user facility.
PNNL Contact
Philip Rasch, Pacific Northwest National Laboratory, Philip.Rasch@pnnl.gov
Funding
This research is based on work supported by the U.S. Department of Energy (DOE) Office of Science, Biological and Environmental Research (BER) program, as part of the Earth and Environmental System Modeling program, and by the Environmental Molecular Sciences Laboratory, a DOE Office of Science user facility sponsored by BER and located at Pacific Northwest National Laboratory. Partial support was provided by the NASA Modeling, Analysis, and Prediction Program and National Center for Atmospheric Research.
Published: May 7, 2021
R. A. Zaveri, et al. “Development and evaluation of chemistry-aerosol-climate model CAM5-Chem-MAM7-MOSAIC: Global atmospheric distribution and radiative effects of nitrate aerosol.” Journal of Advances in Modeling Earth Systems 13, e2020MS002346, (2021). [DOI:10.1029/2020MS002346]