Novel Titanium Dioxide Structures Enhance Photoactivity
Crystal deformation offers a novel design paradigm for photoactive materials.
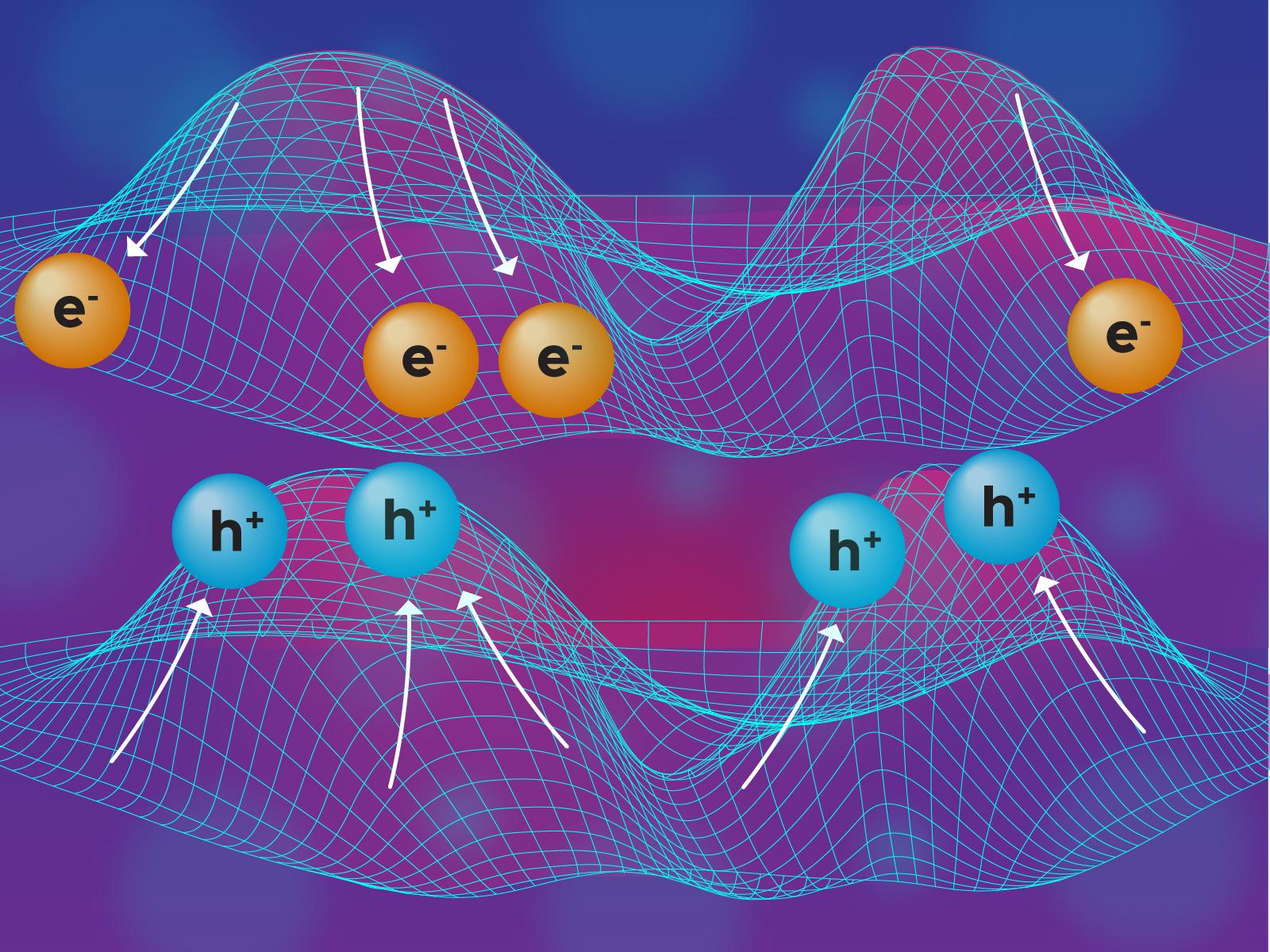
Schematics of the 3-D band edge bending: small atomic displacements from the crystalline lattice sites create internal electric fields with their relative energies.
(Illustration by Stephanie King | Pacific Northwest National Laboratory)
The Science
Titanium dioxide (TiO2) is a naturally occurring compound with a wide range of applications. It is used in green energy materials because of its ability to promote chemical reactions. However, TiO2 suffers from limited photoconversion efficiency and limited ability to transport electrical charge carriers throughout its bulk. Researchers discovered a new type of TiO2 structure that occurs during the transition from one crystal phase to another. This transitional structure contains atoms that occupy continuously altered sites across the bulk, thus creating a gradual gradient in the atomic positions. This “atomic-gradient structure” alters the electronic properties in 3-D, significantly increasing active volume and transporting charge carriers more efficiently than conventional TiO2.
The Impact
A long-standing challenge in the production of photoactive, or sunlight-capturing, materials is the ability to synthesize materials that are highly active throughout their bulk. The discovery of this new crystal structure under tunable conditions indicates that a new design process may be used to produce better energy materials besides TiO2. The potential new design process may include a variety of tunable techniques to produce more efficient crystals using the same cost-effective materials as before. If the photoactivity of these materials can be changed without altering their chemical composition using this heating method, there are likely other such methods that produce similar results.
Summary
TiO2 anatase crystals are used in many green energy and photocatalytic materials. While they are generally considered to be affordable and safe, they exhibit a low active volume across the bulk of the crystals. Heating TiO2-B induces structural changes in the crystals from the TiO2-B phase to anatase, forming continuous atomic positional deformations within them. This change can be induced using established heating techniques, and the precise structure can be tuned by controlling the temperature and duration of heating. These new “atomic-gradient” structures enable the formation of internal electric field gradients that allow the material to become nearly 100% active across its bulk. These post-heat-treatment crystals exhibit up to 5× greater photoactivity than their untreated counterparts. When heated at 550 °C for 6 hours, TiO2-B crystals reach their maximum efficiency.
The transformation of these crystals during the heating process was observed at the atomic scale using in situ transmission electron microscopy (TEM) and corroborated using density functional theory simulations. TEM characterization experiments were performed at EMSL, the Environmental Molecular Sciences Laboratory, a U.S. Department of Energy user facility located at PNNL.
Funding
- U.S. Department of Energy (DOE) Office of Science, Basic Energy Sciences (BES) Program, Early Career Research Program Award
- DOE BES Division of Materials Sciences and Engineering Award
Published: April 12, 2021
P. Ren, et al., “Atomic Gradient Structure Alters Electronic Structure in 3D across the Bulk and Enhances Photoactivity.” Advanced Energy Materials, 2003548, (2021). [DOI: 10.1002/aenm.202003548]