Enhancing Photocatalytic Efficiency Using Charge Carrier Separation in a Layered Heterostructure
Proposing a new synthetic route to layered materials that demonstrate efficient photocatalysis
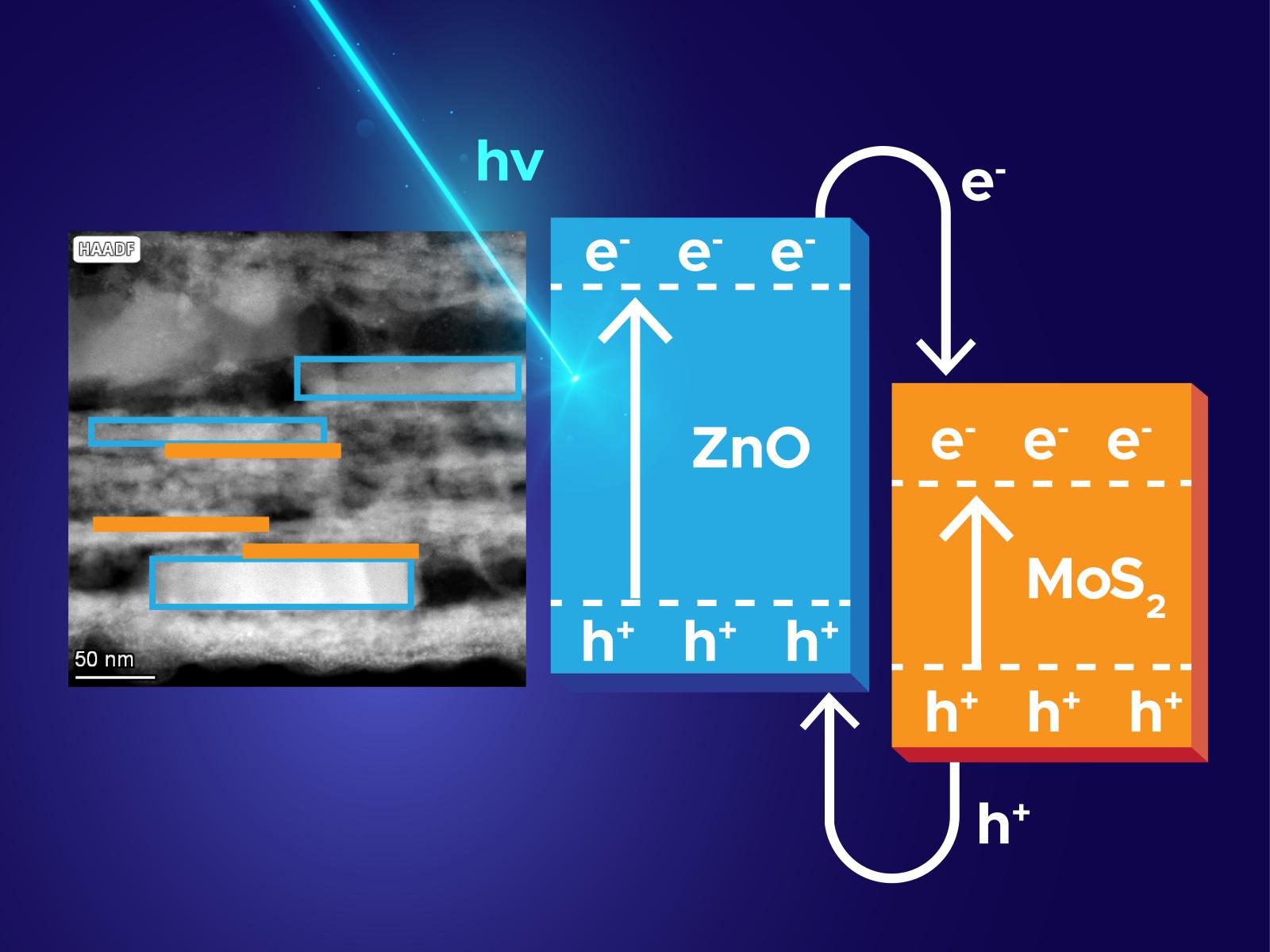
Layering materials creates an electronic structure that leads to physical charge separation after photoexcitation.
(Composite image by Stephanie King and Lili Liu | Pacific Northwest National Laboratory)
The Science
Converting light to electricity requires separation and movement of charges in materials. One approach to make this process more efficient is to build structures with multiple materials that lead to a physical separation of positive and negative charges, reducing the charge recombination. Researchers developed a new solution phase method to create layered heterostructures of zinc oxide (ZnO) and molybdenum disulfide (MoS2). The straightforward synthesis produces heterostructures where single layers of MoS2 are epitaxially layered on multilayered ZnO. This allows charges, positive and negative, to localize on the different materials, creating a physical charge separation. These heterostructures are 50 percent more efficient photocatalysts than individual or physically mixed ZnO and MoS2 materials.
The Impact
Exploiting the emergent electronic and photocatalytic properties of layered heterostructures in different applications requires developing reliable and scalable synthesis approaches. Current vapor deposition-based methods are plagued by poor scalability and are labor intensive. This work developed a synthesis method that leads to well-defined heterostructures of layered materials in a straightforward and simple solution-based process. The scalability of precise synthesis is critical for developing next-generation functional materials for solar cells and other clean energy applications.
Summary
Photocatalysts convert light energy into fuel for chemical reactions through the generation of separated charges. Many inorganic materials have been proposed as candidates for high stability photocatalysis, but the charges formed within these materials often rapidly recombine before they can react. One avenue to creating long lifetimes for charges is by using heterostructures, which contain multiple types of materials, that physically separate positive and negative charges. Researchers developed a new, solution-based method to synthesize a ZnO and MoS2 layered heterostructure. This synthesis produces materials with alternating MoS2 and ZnO layers that are epitaxially attached. The layered structure forms due to the stabilization of the intermediate zinc hydroxide phase on MoS2 surface and its subsequent transformation into the two-dimensional ZnO. Studies examining the degradation of rhodamine B showed that the new heterostructure was 50 percent more efficient at the photocatalytic reaction than either material separately or an unconnected mixture of the two materials. This work represents a step toward developing the scalable, precise synthesis that is critical for producing the next-generation functional materials needed for solar cells and other clean energy applications.
PNNL Contact
Maria Sushko, Pacific Northwest National Laboratory, maria.sushko@pnnl.gov
Lili Liu, Pacific Northwest National Laboratory, lili.liu@pnnl.gov
Funding
This research was performed at Pacific Northwest National Laboratory (PNNL) with support from the Department of Energy, Office of Science, Basic Energy Sciences, Division of Materials Sciences and Engineering, Synthesis and Processing Science program (PNNL FWP 12152). Electron microscopy and computational work used resources of EMSL, the Environmental Molecular Sciences Laboratory, a Biological and Environmental Research program user facility.
Published: January 23, 2024
Liu L., P. Chen, X. Zhang, L. Kovarik, J.J. De Yoreo, J. Liu, M.L. Sushko. “Solution synthesis of two dimensional zinc oxide (ZnO)/molybdenum disulfide (MoS2) heterostructure through reactive templating for enhanced visible light degradation of rhodamine B,” Advanced Composites and Hybrid Materials, 6(6), 223 (2023). [DOI: 10.1007/s42114-023-00780-8]