Advanced Computing, Mathematics and Data
Research Highlights
February 2009
Measurements at the Genome-Scale Made in Nanotoxicity Study
Macrophage cell model gives insight to toxic effects of nanomaterials
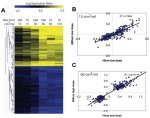
This figure shows the relationship between mRNA abundance changes and particle surface area dose for 404 genes regulated in response to 10- and 500-nanometer amorphous silica particles. Genes shown in the majority pattern A naturally clustered according to the particle surface area dose. In contrast, the genes for patterns B and C continued to separate by particle size and mass dose (data not shown). Correlation analysis for the majority pattern A genes indicated a strong overall correlation between the gene expression ratio and particle surface area dose across all treatment groups. Enlarged View
Results: The increasing use of nanomaterials in products from semiconductors to sunscreen is revolutionizing products in many industries. This popularity results from significant changes in the chemical reactivity and physical properties of many materials significantly change at the nanoscale—that is, having at least one dimension of 100 nanometers or less.
However, the same properties that make nanomaterials attractive for commercial and medical use may also enhance their biological reactivity, raising concerns about their potential toxicity. For example, materials classically considered biologically inert, such as amorphous silica, titanium dioxide and gold have been reported to cause exacerbated biological responses when used at the nanoscale.
Scientists are now closer to determining what a "safe dose" of nanomaterials is to humans, thanks to a comprehensive study recently published by the Pacific Northwest National Laboratory. Using an in vitro model of macrophage cells, they demonstrated that the ability of amorphous silica particles—akin to sand—to stimulate inflammation and toxicity in cells scales closely with the total particle surface area.
"To our knowledge, this is the first study to use genome-scale measurements to evaluate whether the cellular effects induced by nanomaterials are dependent on particle size," said PNNL scientist Katrina Waters, lead author of an article that appeared in Toxicological Sciences. "Our results suggest that the surface chemistry properties of amorphous silica that are responsible for its bioactivity do not significantly change as particle size decreases to the nanoscale."
Why does it matter? The molecular mechanisms by which nanoparticles stimulate cellular responses are still poorly understood. Yet it's clear that particle surface chemistry is an important driving factor. By identifying the early transcriptional events and pathways activated by amorphous silica in macrophages—white blood cells that surround and kill invaders to the tissues—the PNNL team's results signi?cantly extend previous studies. Because of their role in immune surveillance, macrophages are important sentinels for biological response to pathogens.
To identify hazards and assess human risk from nanoparticle exposure, researchers must extrapolate dose-response data from animal studies to human systems. Therefore, accurate interpretation of size-dependent biological responses to nanoparticles depends on the dose metric used for comparison—in this case, particle size. This is because for a given mass of particles, the total surface area increases with decreasing particle diameter, making it an essential determinant of the fraction of reactive groups on particle surfaces.
This study demonstrated that the particle surface properties that dictate biocompatibility of amorphous silica are essentially the same at the nanoscale as a function of surface area. The generalized strategy described in this study can be extended to other nanomaterials both to identify mechanism of action and to prioritize more expensive toxicity studies in animals.
Methods: The PNNL scientists used a genome-wide approach to investigate whether the cellular pathways activated by amorphous silica are size dependent. Amorphous silica was chosen because of its increasingly important role in nanotechnology. It's used in production of high-efficiency photovoltaics and tires, and in consumer products such as drug delivery vehicles, toothpaste, sunscreen, cosmetics and nutraceuticals.
They performed whole genome microarray analysis of the early gene expression changes induced by 10- and 500-nm particles. These showed that the magnitude of change for the majority of genes affected correlated more tightly with particle surface area than either particle mass or number. The researchers also identified particle size-specific gene expression changes. (see figure).
Using advanced bioinformatics approaches, the team was able to demonstrate that the biological pathways represented by the size-specific gene expression changes were nearly identical, irrespective of particle size. Direct comparison of the cell processes represented in the 10- and 500-nm particle gene sets using gene set enrichment analysis revealed that among 1009 total processes represented in the data, none were statistically enriched for one particle size group over the other.
What's next: The key mechanisms involved in silica nanoparticle-mediated gene regulation and cytotoxicity have yet to be established. For example, scientists don't know whether macrophage cell responses are initiated by particle contact with the cell membrane or if receptor binding or particle internalization is required. Such studies should help in developing strategies for enhancing the beneficial properties of nanomaterials while reducing potential adverse health effects mediated through macrophage interactions.
Acknowledgments: The work was sponsored by the National Institutes of Health and internal funding at PNNL. Particle characterization and transmission electron microscopy were done at the U.S. Department of Energy's Environmental Molecular Sciences Laboratory at PNNL. The research team included Katrina Waters, Lisa Masiello, Rick Zangar, Colette Sacksteder, Ruimin Tan, Barbara Tarasevich, Norm Karin, Ryan Quesenberry, Somnath Bandyopadhyay, Justin Teeguarden, Joel Pounds and Brian Thrall. This work is advancing science to achieve predictive understanding of multi-cellular biological systems.
Reference: Waters KM, LM Masiello, RC Zangar, BJ Tarasevich, NJ Karin, RD Quesenberry, S Bandyopadhyay, JG Teeguarden, JG Pounds, and BD Thrall. 2009. "Macrophage Responses to Silica Nanoparticles are Highly Conserved Across Particle Sizes." Toxicological Sciences 107(2):553-569.


