Advanced Computing, Mathematics and Data
Research Highlights
February 2009
Water, Water Everywhere
New model improves calculation of electric fields
Results: One of the toughest challenges in molecular simulation is to construct computational models that contain enough of the right physics to produce results scientists can trust. Pacific Northwest National Laboratory scientists Dr. Shawn Kathmann and Dr. Christopher Mundy and colleague Dr. I-Feng William Kuo of Lawrence Livermore National Laboratory have solved part of that problem. They have produced the first model to faithfully depict the electric field present at the point, or interface, where the liquid and vapor forms of water meet.
The model also comes closer than other simulations to matching calculations derived from experiment. Details are on the Web in the Journal of the American Chemical Society.
Why it matters: "In molecular simulation, a model is only as good as the physics that goes into it. More physics leads to better models," Kathmann said. "The effects of electrons and how to include them in models can make a big difference in the accuracy of a model," he said. "We found that you have to consider the electrons individually if you want an accurate representation of an electric field."
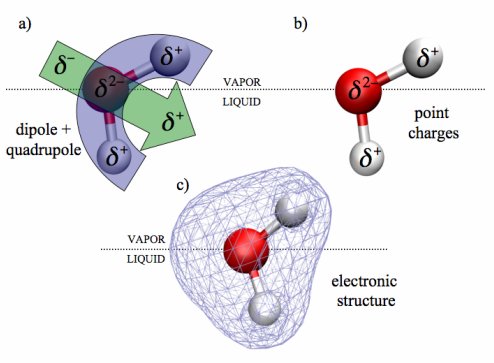
Unlike previous models (top), a new computational model (bottom) includes the full “cloud” of electrons in a water molecule.
The new model provides an improved tool for acquiring that information. The model calculates the molecules' full electronic charge distribution—all of the water electrons and how they interact with other electrons. The type of information this model can provide is significant in many areas of chemical physics research, including energy, biology, materials science and global warming.
Methods: Scientists know that the dominant interaction among molecules is electrical and that very interesting chemistry occurs at interfaces, where one form of matter meets another. From electricity on a child's balloon that makes your hair stand up, to electron transport in transistors, electric fields are what make things move. Yet calibrating an electric field is immensely complex, even for a molecule as simple as water.
Surface potential is the term given to the measurement of an electric field at an interface. Scientists use surface potential calculations to describe and predict chemical reactions at a vapor-liquid interface. Many scientists have attempted to determine the surface potential of the vapor-liquid interface of water. But, they do not agree on the importance of the field or if the charge distribution is positive or negative.
The primary obstacle has been lack of computing power. The high-performance computing resources enabled the research team to apply quantum physics to the problem. These resources are located at Livermore and the U.S. Department of Energy's EMSL, a national scientific user facility at PNNL.
The new model revealed that the strength of the electric field is much smaller than previously estimated and that the charge is always negative.
The team started by calculating the behavior of the electronic cloud that envelops every water molecule at the vapor-liquid interface. In water that is either all liquid or all gas, it is the intermingling of these electronic clouds that holds the molecules together. However, electrons behave differently where the liquid and vapor meet. This interface is a sort of no-man's-land, where electrons move from one state to the other and back again, aiding or restricting chemical processes and other molecular interactions.
The team then compared the new model with previous models and experiments. Earlier models viewed the electrons in water as set along certain pathways or gathered at the nuclei of the atoms. Dealing with the electrons as a cloud confirmed predictions that the density of the electrons significantly influences the electronic properties of the vapor-liquid interface of water.
What's next? Future studies will seek to further quantify explicit inclusion of the electronic charge cloud. For example, researchers will address the surface potential and electric field at the interface between a salt crystal and liquid water, as well as electronic effects on the reaction path of various ions through the vapor-liquid interface of water.
Acknowledgments: The DOE Office of Basic Energy Sciences, Chemical Sciences Program funded the research. Shawn M. Kathmann, I-Feng William Kuo, and Christopher J. Mundy used the computing resources located at DOE's Lawrence Livermore National Laboratory and at EMSL.
The work supports the Laboratory's mission to strengthen U.S. scientific foundations for innovation by developing tools and understanding required to control chemical and physical processes in complex multiphase environments.
Reference: Kathmann SM, IFW Kuo, and CJ Mundy. 2008 "Electronic Effects on the Surface Potential at the Vapor-Liquid Interface of Water." Journal of the American Chemical Society, 130:16556-16561.
